ff
Crystal Engineering for Green Chemistry
Crystal engineering lends itself to green chemistry practices because reactions carried out within a crystal limit the use of toxic organic solvents, which are generally not required (i.e., solvent-free). These reactions are atom-economical (i.e., all reactants are converted into products) and highly selective (i.e., no by-products). The energy required for the reactions can be supplied by sunlight (i.e., ultraviolet), and the components that direct the reactions are recyclable templates and catalysts (i.e., similar to biochemical enzymes).
Each of these features aligns with the 12 Principles of Green Chemistry. In our research, we are also developing methods to carry out solid-state reactions through mechanochemical and thermochemical processes, which may enable large-scale production of materials while continuing to limit the use of toxic solvents.
Our goal is to control chemical reactions in organic crystals. Although reactions in crystals have been known for over a century, the ability to use the solid state in the same way as the liquid phase to routinely synthesize organic molecules has not yet been realized. This limitation stems from the lack of a general method capable of assembling a wide variety of reactants to undergo reactions in the solid state.
What this has meant is that the true capacity of organic crystals to generate a variety of molecules—and the many green chemistry benefits that come with it—has remained largely unrealized.
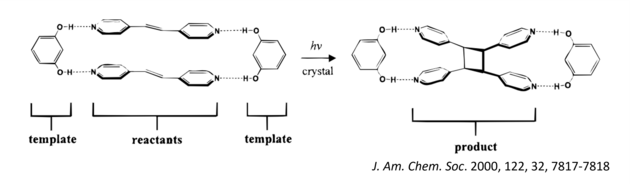
Our group has already shown that cocrystallization (i.e., the process of combining molecules) of 1,3-benzenediol, or resorcinol, with a bis(pyridyl)ethylene produces a multicomponent crystal composed of two different molecules, or a binary cocrystal, in which the alkenes are positioned by hydrogen bonds to undergo a photodimerization reaction.
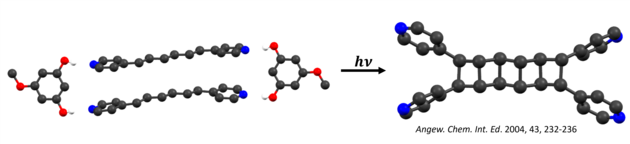
Irradiation of the cocrystal with ultraviolet (UV) light—either from laboratory sources or from sunlight—generated a tetra(pyridyl)cyclobutane (CB) as the sole product, in quantitative yield and in gram quantities. The key to this method is that the template molecules isolate the reactants from the effects of molecular packing. Furthermore, the template molecules can be easily recovered and recycled. Our group has further expanded this method to produce architecturally and energetically rich CBs such as ladderanes and cubanes. The overarching idea is to use organic crystals as green chemical laboratories that provide degrees of synthetic “freedom” typically realized in solution.